Introduction
This guideline provides a comprehensive overview of the types of research Mohawk Valley Health System (MVHS) personnel (faculty, fellows, residents, nurses etc.) may undertake, the necessary CITI training modules, and when IRB approval or a research protocol is required. The goal is to ensure that all research conducted is ethical, compliant with institutional policies, and protects the rights and privacy of participants.
General Research Questions
What’s the definition of Investigator Initiated and Sponsored Research?
Investigator Initiated Research (IIR) refers to a study that the investigator creates the question, develops a hypothesis, protocol and is overall responsible for execution. Sponsored Research refers to a research question and study that is created by the government, a federally funded agency, private industry, or a pharmaceutical company. Please note, that although the descriptions of different types of studies and types of training are listed below; study Sponsors have the final determination on what training and regulatory requirements are necessary for participation.
What research training is necessary?
The type of training required, depends on the nature of the research study. Documented protocol training of the study staff is required. This demonstrates that everyone on the study has read the protocol, has a certain level of understanding of the study, and is prepared to execute what is needed to complete the study.
Another required training module is the “Collaborative Institutional Training Initiative (CITI) platform (https://about.citiprogram.org). This website hosts various self-paced training material with an assessment required at the end. There is numerous training material present that can be personalized based on the type of study you will be participating in and your role. Additional information regarding what training is appropriate for role within the study is listed in subsequent sections.
What is a protocol? Do I need one?
A protocol is a document that outlines the research question and the steps needed to answer that question. In IIR studies, this may be created by the principal investigator (PI) or other members of the study team. In Sponsored studies, the protocol is created and distributed by the Sponsor. The protocol acts as reference document for the study team and ensures that the study is executed properly. Important elements of the protocol include the research question (hypothesis), study goals (objective), how the study will be conducted (procedures), and who will be included (study population and inclusion/exclusion criteria). Additional elements may include statistical considerations (how many people will participate to get the needed data and how will this data be analyzed) and safety elements (description of risk and any anticipated adverse events).
Yes, you do need a study protocol. The presence of a protocol will ensure that the study goals are met and the study question is answered completely. The protocol will also serve as a training guide for study members and ensure study procedures are consistently implemented across all study personnel and subjects. This study protocol will be used by the Research Department /Clinical Trials Office (CTO) to determine the classification of study, what training will be necessary, and any additional resources may be needed; and the Institutional Review Board (IRB) will determine what submission and informed consent is needed (if any). If you do not have a study protocol, questions from the feasibility questionnaire should help generate the necessary components. If you have further questions regarding the protocol, please contact the Research Department/CTO.
Do I need funding for this research study?
Sponsored studies (conceived and operated by external organizations) will most likely have funding and provide you with a study budget that will break down the study finances. IITs may have an organization is funding the study, but most IITs do not have funding. Funding for your study may be relevant if you need additional storage (documents, samples, etc.), a statistician, or additional resources you do not currently have. If you have a concern regarding whether funding is needed for your project, please contact the Research Department/CTO.
Who can lead a research study?
Any appropriately credentialed research trained MVHS faculty/personnel can submit an application to lead a research study as a Principal Investigator (PI). The PI has the ultimate responsibility for the conduct and execution of the study and the personnel working on the study. Residents and fellows may not act as a PI of a study at MVHS. To submit and execute a study, they will need to collaborate with current faculty in good standing.
Types of Research Studies
Research studies may be classified several ways and sometimes terminology may overlap. The information below classifies studies in two different ways: how the data is collected (prospective vs retrospective) and the intent of the study. Each type of study has unique requirements and implications for IRB approval, informed consent, and CITI training. For more specific details regarding CITI training, please see the section below. If you have any questions, regarding what classification aligns with your research, please contact the Research Department /CTO. Please note that if you participate in a Sponsored study, the Sponsor may have different guidelines for educational requirements and regulatory submissions.
How data is collected:
- Retrospective Studies (how is the data collected)
- Definition: Studies that analyze existing data, such as medical records, to identify trends, correlations, or outcomes related to past interventions.
- When to Use: When investigating outcomes or associations using previously collected data.
- CITI Modules Required:
- Human Subjects Research – Biomedical or Social-Behavioral-Educational
- Responsible Conduct of Research (RCR)- Biomedical or Social/Behavioral
- Protocol/IRB Approval: Required
- Informed Consent: May be waived if the study meets certain criteria (e.g., minimal risk and no direct patient contact).
- Prospective Studies (how is the data collected)
- Definition: Studies designed to follow participants over time to observe the effects of certain exposures or interventions.
- When to Use: For collecting data about outcomes that occur after the study begins.
- CITI Modules Required:
- Human Subjects Research – Biomedical or Social-Behavioral-Educational
- Responsible Conduct of Research (RCR)- Biomedical or Social/Behavioral
- Good Clinical Practice (GCP)
- Conflicts of Interest in Healthcare
- Protocol/IRB Approval: Required
- Informed Consent: Required
Intent of the study:
- Case Report
- Definition: A detailed account of a 1-2 patient’s clinical presentation, diagnosis, treatment, and follow-up, focusing on unique or novel aspects.
- When to Use: When documenting a rare or novel occurrence that could inform clinical practice or generate hypotheses for future research.
- CITI Modules Required:
- Human Subjects Research – Biomedical or Social-Behavioral-Educational
- Responsible Conduct of Research (RCR)- Biomedical or Social/Behavioral
- Protocol/IRB Approval:
- Typically, Not Required: If the report involves fewer than three cases and no identifiable information is used. This study may still be required to be submitted to the Research Department /CTO and Graduate Medical Education (GME) office to ensure that there are no duplicate studies and the study is appropriately tracked.
- Informed Consent:
- Required if identifiable patient information is included.
- Case Series
- Definition: A study that aggregates and analyzes data from three or more patients with similar conditions or treatments.
- When to Use: When assessing patterns, outcomes, or responses in a group of patients, often used in early-stage research.
- CITI Modules Required:
- Human Subjects Research – Biomedical or Social-Behavioral-Educational
- Responsible Conduct of Research (RCR)- Biomedical or Social/Behavioral
- Protocol/IRB Approval: Required
- Informed Consent: Required if identifiable patient information is included.
- Quality Improvement (QI) Projects:
- Definition: Projects aimed at improving healthcare processes, outcomes, or efficiencies within a specific setting, often without the intent of contributing to generalizable knowledge.
- When to Use: For internal evaluations of clinical practices, workflows, or patient outcomes.
- CITI Modules Required:
- Human Subjects Research – Biomedical (if patient data is involved)
- Responsible Conduct of Research (RCR)- Biomedical or Social/Behavioral
- Conflicts of Interest in Healthcare (if applicable)
- Protocol/IRB Approval: Not required for internal projects, but IRB review may be needed if results will be published or if the project could be considered research.
- Informed Consent: Usually not required for QI projects but consult with the IRB if there’s any doubt.
- Post-Approval Studies or Phase IV studies
- Definition: Studies conducted after a treatment, drug, or device has received regulatory approval, often for monitoring long-term safety and effectiveness.
- When to Use: For ongoing evaluation of interventions in a real-world setting.
- CITI Modules Required:
- Human Subjects Research – Biomedical or Social-Behavioral-Educational
- Good Clinical Practice (GCP) – FDA-Regulated Research
- Conflicts of Interest in Healthcare
- Responsible Conduct of Research (RCR)- Biomedical or Social/Behavioral
- Protocol/IRB Approval: Required
- Informed Consent: Required
- Registries
- Definition: A systematic collection of data about patients with a specific condition or treatment, used to track outcomes over time.
- When to Use: For long-term tracking of patient outcomes or to monitor the effectiveness of treatments.
- CITI Modules Required:
- Human Subjects Research – Biomedical or Social-Behavioral-Educational
- Responsible Conduct of Research (RCR)- Biomedical or Social/ Behavioral
- Conflicts of Interest in Healthcare
- Protocol/IRB Approval: Required
- Informed Consent: Required
- Phase I, II and III Studies
- Phase I studies focus on safety in a small population of healthy participants. Patient safety is prioritized during these ‘first in human studies’.
- Phase II studies have a larger population participating than phase I studies, but all participants are affected by the condition of interest. Patient safety and efficacy are prioritized, as well as finding an appropriate dose/treatment regimen for that population.
- Phase III studies may involve hundreds to tens of thousands of affected subjects. The treatment of interest may be compared to similar products/treatments on the market. Patient safety and efficacy is prioritized.
- CITI Modules Required:
- Human Subjects Research – Biomedical or Social-Behavioral-Educational
- Responsible Conduct of Research (RCR)- Biomedical or Social/ Behavioral
- Good Clinical Practice (GCP)
- Conflicts of Interest in Healthcare
- Protocol/IRB Approval: Required
- Informed Consent: Required
CITI Training Requirements
CITI training is mandatory for MVHS personnel that are creating, participating or assisting in the execution of research studies. The specific modules depend on the type of study being conducted, your role in the research study, and the nature of the protocol (some studies may require additional training for sample collection and/ or shipping).
CITI modules can be accessed at (https://about.citiprogram.org). To review and obtain the necessary courses, please affiliate with MVHS. If you have previously completed CITI courses with another institution, you do not need to create another login. Please login with your previous credentials and affiliate with MVHS. Once you log into the system and associate yourself with MVHS, please select which courses are relevant to your study. You will then narrow down your selection, when you specify your role.
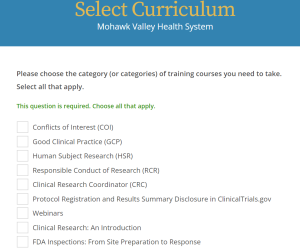
Below are the core and specialized modules (and how they appear in the CITI module):
Core Modules:
- Human Subjects Research – Biomedical or Social-Behavioral-Educational
- Conflicts of Interest in Research

- Responsible Conduct of Research (RCR)- Biomedical or Social/Behavioral

Specialized Modules (based on study type):
- Good Clinical Practice (GCP)

The Research Study Submission Process
For internal research projects and IIT studies:
Please submit the MVHS internal research projects/IIT feasibility form. This form will review the study details to determine if:
- There are appropriate resources to complete your study
- There is no duplicate or competing study
This will also assist the Research Department /CTO with routing your study for appropriate IRB submission and will assist with the creation of your protocol/ study resources (if needed).
For sponsored research:
Please submit the MVHS Sponsored research feasibility form. This form will review Sponsor materials to ensure:
- There are appropriate resources to complete your study
- There is no duplicate or competing study
- All necessary documents have been received to proceed with study submission.
Please note that no study will be reviewed by the IRB for approval, unless there is accompanying documentation from the CTO.
Informed Consent Considerations
Informed consent documents are an important study tool used to engage a potential research subject, educate them about the study, let them know what is expected of them, and give options for participation. The document will also detail what information of theirs will be shared and who will look at it (HIPAA). Language in informed consent documents must be well prepared to explain necessary information, clearly and concisely. We currently have an informed consent template that can be modified for use in your study (if not already provided by the Sponsor). During this process if you have any questions or concerns, please contact the CTO.
Institutional Resources and Support
IRB Office: Contact information and resources for assistance with protocol submissions and IRB queries.
Research Office: Support available for research design, data analysis, and publication.
Appendices
Appendix A: Flowchart for IRB Submission Process
Appendix B: Investigator Initiated Trials/ Internal Research Projects Feasibility Form
Appendix C: Sponsored Trials/ Research Projects Feasibility Form

